A recent randomized controlled trial of varenicline (Chantix) combined with psychological support has significantly increased the chance of quitting smoking for 300 smokers with type 2 diabetes mellitus
Smoking can deeply affect people with type 2 diabetes mellitus (T2DM). The combination of hyperglycemia and inhalable toxicants in cigarette smoke can accelerate progression of disease complications (e.g. coronary heart disease, stroke, peripheral arterial disease, retinopathy and nefropathy).
Moreover, these patients have unique behavioral and metabolic effect that should be considered when addressing smoking cessation in T2DM; for example smokers with diabetes can experiment a lower motivation to quit due to the risk of weight gain post cessation increasing the risk of relapse.
Nonetheless, many studies have shown better glycemic control and lowered cardiometabolic risk factors after stopping smoking and helping T2DM smokers quit is a top priority.
Behavioral counseling and smoking cessation medications are generally combined to help people quit smoking, but the evidence for efficacious cessation interventions in patients with diabetes is limited. In particular, efficacy and safety of varenicline (Chantix) for smoking cessation in smokers with diabetes has not been extensively studied.
A randomized controlled trial conducted by researchers of CoEHAR, Center of Excellence for the acceleration of HArm Reduction at University of Catania and Mossakowski Medical Research Institute of Poland, has now evaluated efficacy and safety of varenicline (Chantix) in type 2 diabetes mellitus patients intending to quit.
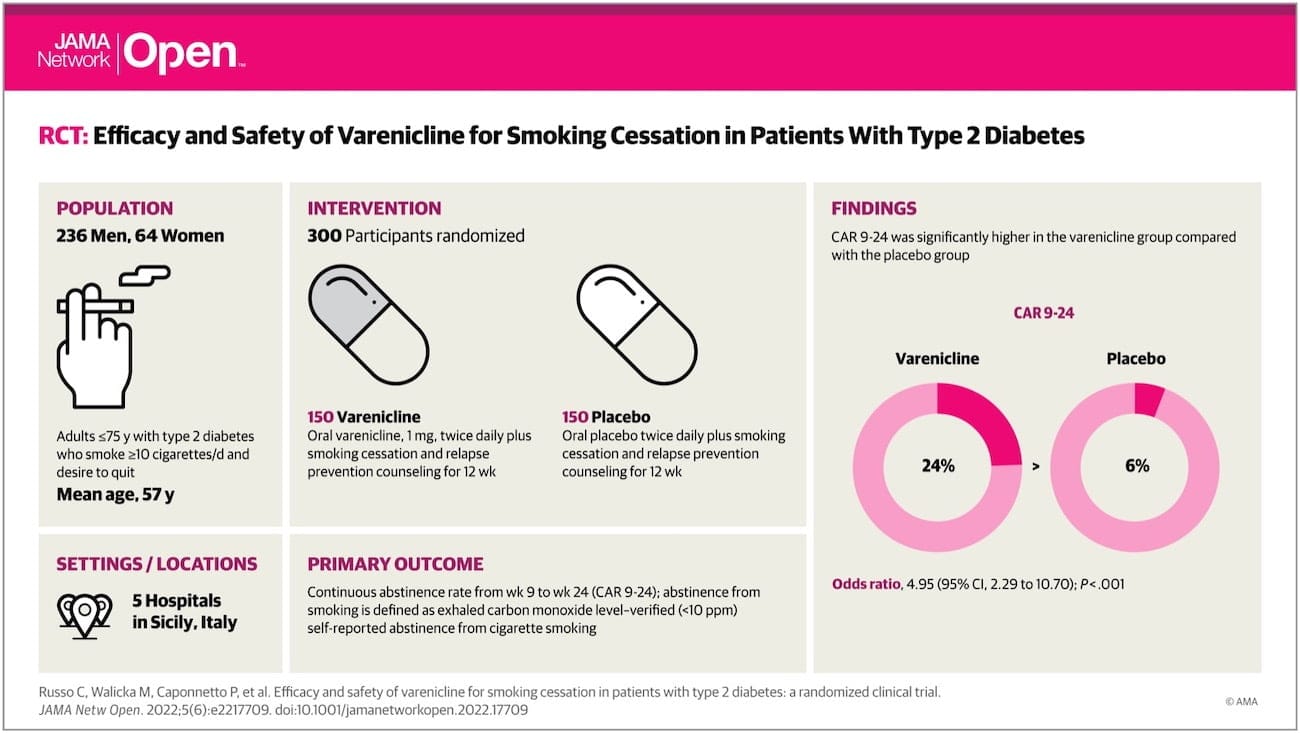
The study “Efficacy and safety of varenicline for smoking cessation in patients with type 2 diabetes: A multicenter double-blind randomized placebo-controlled trial”, first in its kind, revealed that varenicline was significantly efficacious at 12, 24 and 52 weeks compared to placebo. Only minor treatment-related adverse events were noted.
“Achieving long term abstinence is a goal that every smokers should have in mind, and more so smokers with diabetes” stated Dr. Cristina Russo, lead author of the study “Smokers with diabetes are aware that smoking increases the chances of cardiovascular and vascular damage and kidney failure but very often cannot stop on their own. We must help these smokers to stop smoking by offering professional advice and most effective treatment. We are glad to show that varenicline, combined with cessation counseling, successfully helped a significant number of patients with diabetes to quit their cigarettes”.
THE STUDY
The study involved a population of type 2 diabetes mellitus patients who were smoking at least 10 cigarettes a day: a total of 300 participant were recruited from five different hospitals in Catania.
The research aim was to assess the efficacy and safety of varenicline compared to placebo for smoking cessation. Once assed the medical and the smoking history, patients received smoking cessation counseling and were divided into different groups: one using varenicline, the other a placebo.
All patients were follow up on a weekly basis during the following 12 weeks after the first visit.
After the treatment phase, patients were invited to clinics at week 13, 24 and 52 to evaluate data on smoking abstinence and treatment condition.
A total of 215 participants in the varenicline and placebo study arm completed the treatment period (73.3% vs 70.0%, in the varenicline and placebo study arm respectively); for the non-treatment phase, 201 participants completed the 24-week study visit (66.6% vs 67.3%, in the varenicline and placebo study arm respectively), and 195 the 52-week study visit (66.6% vs 63.3%, in the varenicline and placebo study arm respectively.
The 7-day point prevalence of smoking abstinence was also significantly higher for the varenicline group than for placebo group at each time point.Type 2 diabetes mellitus smokers in the varenicline group were at least three times more likely to quit compared to the placebo group throughout that study.
Regarding adverse effect of varenicline treatment, most of them were rated as mild or moderate. The most common ones leading to treatment discontinuation were anxiety and depression. The most frequent occurring in the varenicline group compared with the placebo group were nausea (27.3% vs. 11.4%), insomnia (19.4% vs. 12.7%), abnormal dreams (12.7% vs. 3.4%), anxiety (11.4% vs. 7.3%) and irritability (9.4% vs. 5.4%)
The safety profile of varenicline in T2DM was similar to that observed in the previous varenicline trials in smokers in the general population. No differences between or within treatment groups in metabolic and cardiovascular parameters were noticed. Notably, no weight gain was observed in the study.
Smokers affected by diabetes should have access to efficacious and safe quitting methods. The longer the smoking habits is, lower the probability of smoking abstinence over time. Varenicline and psychological support can minimize the risk od diabetes and lead to higher chances of cessation. Besides, the most common side effect of smoking cessation in diabetes patients, weight gain, has not been observed in the study.
Data from the research provided evidence to support varenicline in smoking cessation programs and to help stop smoking in diabetes education programs.